Regional committees for medical and health research ethics (REC)
Pre-approval from REC
The regional committees for medical and health research ethics must give pre-approval:
- Medical and health research projects
- General research biobanks
- Exemption from confidentiality for other types of research
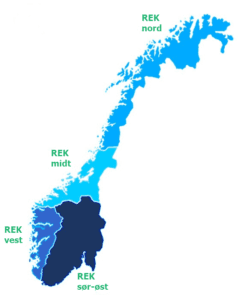
General inquiries should be directed to the REC in the geographical region to which you belong.
Inquiries about submitted applications and cases should mainly be made via the portal or directed to the REC region that processes the case.
E-mails should be sent to post@helseforskning.etikkom.no with the name of the relevant REC/committee in the subject field (REK sør-øst A/B/C/D, REK vest, REK midt or REK nord)
The committee secretariats can be contacted by phone or e-mail. Meetings with the secretariat/case officer should be arranged in advance. See information for the individual REC and secretariat in the individual tabs.
- REK south-east: Agder, Vestfold and Telemark, Viken, Oslo, Innlandet
- E-mail: rek-sorost@medisin.uio.no
- REK west: Rogaland, Vestland
- E-mail: rek-vest@uib.no
- REK Central: Møre and Romsdal, Trøndelag
- E-mail: rek-midt@mh.ntnu.no
- REK North: Nordland, Troms and Finnmark, Svalbard
- E-mail: rek-nord@asp.uit.no